Vapour Pressure

Henry’s Law describes the relationship between the solubility of a gas in a liquid and the partial pressure of that gas above the liquid surface. A range of experimentally determined Henry’s constants are tabulated and can be used to determine the solubility of various gas species in water.

Raoult’s law gives a method of estimating the composition and pressure of the vapour above a liquid mixture. This article describes the basis of Raoult’s law and provides an example of how to apply it.
Dalton’s law provides a method by which the total pressure of a gas mixture can be calculated using the partial pressures of the component gases of the mixture.
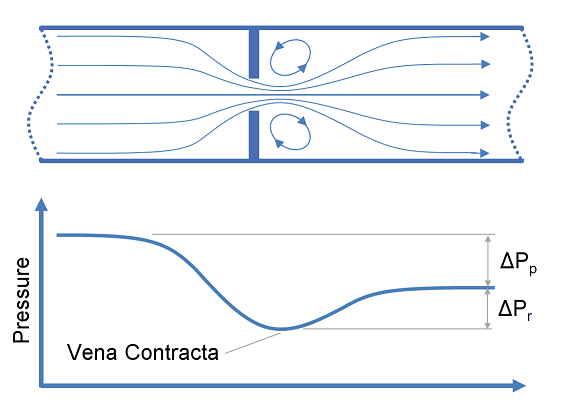
Restriction orifices and control valves are commonly used for pressure reduction and measurement of flow rates, however for a liquid system, excessive pressure drop across these items of equipment may result in cavitation. This article describes methods of predicting cavitation across restriction orifices and valves and proposes designs which may be used to avoid cavitation.
Fittings such as elbows, tees, valves and reducers represent a significant component of the pressure loss in most pipe systems. This article discusses the differences between several popular methods for determining the pressure loss through fittings. The methods discussed for fittings are: the equivalent length method, the K method (velocity head method or resistance coefficient method), the two-K method and the three-K method. In this article we also discuss method for calculating pressure loss through pipe size changes as well as control valves.
Cavitation is the formation and subsequent collapse of vapour bubbles in a flowing liquid and is often responsible for significant damage flow equipment such as pumps. This article will provide an overview of cavitation and provide insight into the identification and prevention of cavitation conditions through the calculation of Net Positive Suction Head (NPSH).