Thermodynamics
When examining thermodynamic processes some simplifying assumptions may be applied to help describe and analyse a given system. These simplifications can be viewed as ‘ideal’ thermodynamic processes and include adiabatic, isenthalpic, isentropic, isobaric, isochoric, isothermal, isentropic, polytropic and reversible processes. This article provides a brief overview of each process type and suitability to a given thermodynamic system.
The laws of thermodynamics are fundamental laws which describe the behaviour of heat and work in a thermodynamic system. These laws forbid phenomena such as perpetual motion machines, a hypothetical machine the development of which was pursued during the industrial revolution.
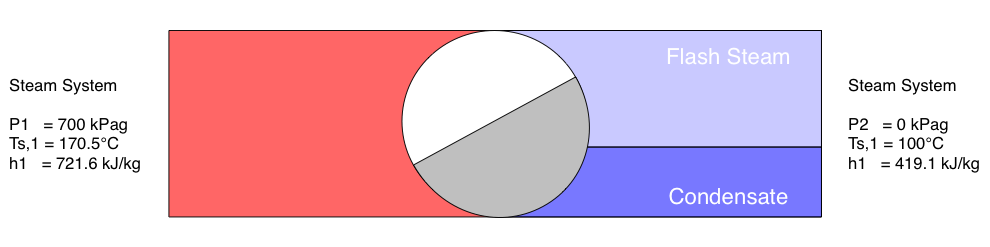
When condensate is discharged from a high pressure steam system to a system of lower pressure, a proportion of the condensate may vaporise to form low pressure steam. This is known as flash steam and may be utilised in low pressure heating systems to improve plant efficiency. This article discusses the generation of flash steam and presents a method by which it can be quantified.